For clinical trial budget developers new to the UK, understanding the time-based costing model is key to interpreting site costs correctly.
Similarly to French budgets, UK budget templates are developed by local teams using a national costing system called iCT (Interactive Costing Tool) and must comply with specific regulatory requirements.
UK clinical trial budgets typically contain two main sheet types: Main and Setup & Closedown. The Main tab includes per-patient costs and additional invoiceable items, while the Setup & Closedown tab captures administrative fees.
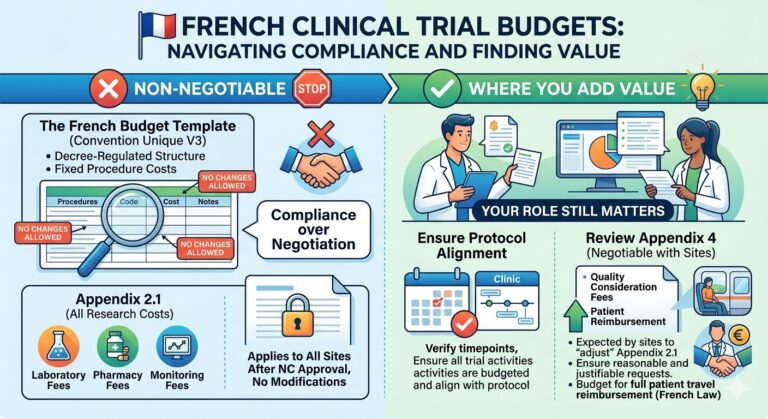
The National Health Service (NHS) mandates that clinical budgets are built on a time-based model. Each procedure is assigned a specific amount of time, depending on the staff involved. As a result, you may see two or more line items for the same procedure. Each line reflects the time contribution of a different staff role (see Column F). In addition, UK budgets are built strictly per protocol and rarely allow costs to be bundled. For example, procedures such as physical examinations and vital signs are listed separately. In the example below, both lines relate to the same procedure (Medical History) but involve different site personnel and specific time allocations.

Per local regulation, there are a number of additional costs (uplifts) added to each line, which normally make UK budgets much higher than the initially anticipated amounts. These additional costs are: Indirect costs (70%, only applicable to the Main tab), Capacity building (20%) and MFF (Market Forces Factor, varies from site to site and depends to a large part on the location of the hospital). It might be a good idea to have a version where these lines are added in order to estimate what the final total cost per patient (TCPP) might be. In the example below you can see that the “real” cost per patient is almost double of what can initially be estimated:

As with France, there is a central authority involved in the review process. In the UK, the National Contract Value Review (NCVR) reviews budget templates and agrees on the resources and pricing required to set up commercial research studies within NHS providers. Once agreed, the budgets are distributed to participating sites, which generally have limited flexibility to request changes.
As you are already an expert on French budgets, let’s compare France vs UK clinical trial budgeting:
| Area | France | United Kingdom |
| Who builds the template | Local country team using a mandatory national template | Local teams using the national iCT (Interactive Costing Tool) |
| Flexibility of structure | Very low – structure and wording are fixed by decree | Low – structure driven by iCT and NHS requirements |
| Costing approach | Fixed fees per procedure | Time-based costing per staff role and procedure |
| Main budget components | Appendix 2.1 (non-negotiable) and Appendix 4 (limited site-level adjustments) | Main tab (per-patient and invoiceable items) and Setup & Closedown tab (administrative fees) |
| Negotiation model | Centralized via National Coordinator (NC) site | Centralized via National Contract Value Review (NCVR) |
| Site-level negotiation | Limited to Appendix 4 | Very limited once NCVR values are agreed |
| Typical cost drivers | Mandatory national rates; site-specific fees | Staff time, indirect costs, capacity building, and Market Forces Factor (MFF) |
| Why budgets look high | All research costs must be included upfront | Mandatory uplifts can significantly increase the final CPP |
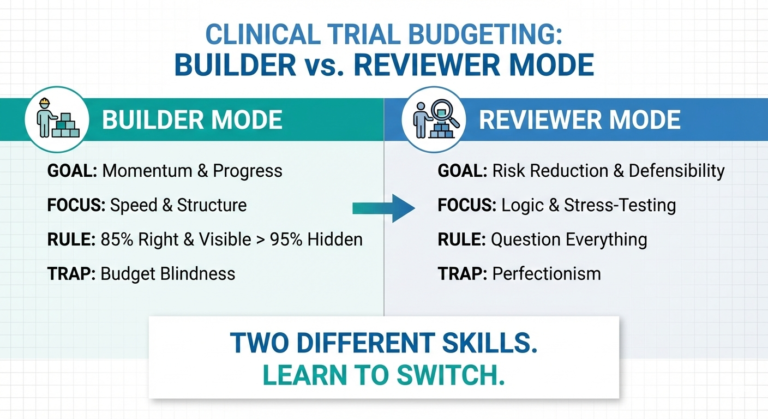
| Your main role as a builder | Ensure protocol alignment and review Appendix 4 for reasonableness | Make sure that there are no “crazy” time assumptions. Inform the sponsor of uplifts |
You can download a 1-image summary here:
Find out who I am and what I am fighting for.
If you agree, disagree, or think this is nonsense, email contact@clinicalbudget.com
If you’d like me to cover a specific topic, you can suggest it here.