Navigating the UK iCT: A Guide to NHS Clinical Trial Budgeting (with Key Differences vs France)

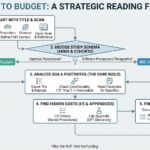
Understanding UK clinical trial budgets starts with the time-based costing model. Unlike fixed-fee systems, each procedure is broken down by staff role and time allocation, often resulting in multiple line items for a single activity. Combined with mandatory uplifts such as indirect costs, capacity building, and Market Forces Factor (MFF), this structure can significantly increase the true cost per patient compared to initial estimates.