If you are new to clinical budgeting, working with the French budget template can feel overwhelming. This guide explains how the system works and where your role still matters.
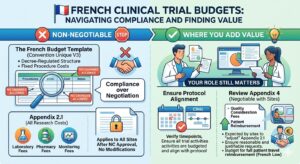
The French budget is in a league of its own – it is less about negotiations and more about compliance. Its structure and wording are regulated by decree and are non-negotiable. You will also notice that you have no influence over the prices for individual procedures. For this reason, French budgets are drafted by the local team, and your involvement as a budget builder is limited.
Negotiations are handled differently than other countries. Initial budget negotiations are handled by an National Coordinator (NC) site and once all approvals are received the budgets are distributed to the other sites that will be participating in this study. It is essential that you understand the differences between the two main tabs of the French budget.
- Appendix 2.1 – Once approved by the NC site, this appendix applies to all other sites and no changes are permitted.
All research-related costs are included in this appendix (laboratory fees, pharmacy fees, SU fees, audit fees, monitoring fees, etc.).
- The budget grid must include costs per procedure and an overall estimate of the total cost per patient; these are not negotiable.
- Titles and descriptions of texts in the matrix cannot be modified. No lines can be added, except for the headings: listed acts, unlisted acts, other costs, and additional costs attributable to research.
- None of the existing lines or wording in the template can be removed (even if not used/applicable for the study)
- If some sections are not applicable (imaging for example), a grey background is applied (recommended).
At the bottom of the Appendix 2.1 tab, summary tables provide an estimate of the per-patient cost and administrative fees per module and in total. These figures should be treated as indicative only, as many fees are paid on a pro rata basis or only if needed and are therefore not included in the total.
- Appendix 4 – Negotiable with each site
This appendix includes the so-called Quality Consideration fees. These fees are comparable to PI fees found in standard budgets. While they are theoretically optional, in practice they are expected by sites; if they are not offered, the study will quickly become less competitive. Sites use this section to “adjust” the Appendix 2.1 fees without directly modifying them, which is not permitted, as mentioned above
Another key point is patient reimbursement. Under French law, once a subject is enrolled, all patient travel costs must be fully reimbursed, regardless of the amount. As stated in Article L1121-11 of the Public Health Code, research involving human subjects must not provide any direct or indirect financial compensation, other than reimbursement of expenses incurred.
“That’s all fine and good, but where do I come in?” I hear you asking, given that almost everything in the French budget is non-negotiable.
In practice, you do have a role to play. Your main responsibility is to ensure that the timepoints included in the budget align strictly with the protocol. The example below shows how visits are typically represented in French budgets and what you need to pay attention to.
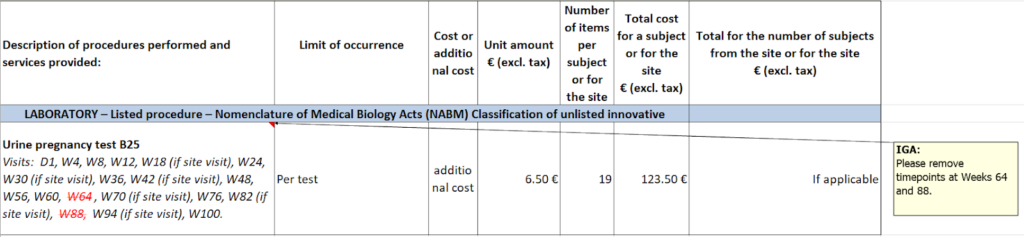
In addition, it is good practice to review carefully any fees added in Appendix 4 and assess whether they are reasonable and justifiable. In one study, the PI requested a fixed amount in EUR as a condition for participation (effectively a “reputation fee”), which required additional discussion and documentation before it could be accepted. On this note, it is one of the countries where donations can be accepted, more information on that can be found in the links at the end of the article.
Below is a summary of the steps for budget development in France:
- The local team creates the French budget based on the mandatory template and rates.
- Once drafted, reviewed, and approved by the Sponsor, the budget is sent to the NC (National Coordinator) site for review.
- The NC reviews the budget and may make additional changes based on local practice and/or needs.
- The NC version of the budget is sent to Sponsor for final review and approval.
- Once approved, this version becomes the final country template and is distributed to the other sites. These sites must accept Annex 2.1 as agreed; any requested changes must be reflected in Annex 4.
For reference, the relevant regulatory framework includes:
- The French Sole Agreement (called “ Convention unique”) framed by French law (order of 16th November 2016 related to the sole agreement and decree n° 2016-1538 of 16th November 2016 as modified by Order on 28 March 2022).
- The current version is the Third version (French Sole Agreement V3 called “Convention Unique V3” or “CUV3”) implemented on 10th April 2022 by the publication in the authenticated electronic official journal n° 0084 on 09th April 2022.
Légifrance – Publications officielles – Journal officiel – JORF n° 0084 du 09/04/2022 (legifrance.gouv.fr)
- (2) different templates of budget and CTA : Coordinating and Associated sites templates provided by the MoH (DGOS) are available on the MoH website :
La convention unique – Ministère de la Santé et de la Prévention (sante.gouv.fr)
Donations: https://www.legifrance.gouv.fr/codes/article_lc/LEGIARTI000042004319?utm_source=chatgpt.com
Find out who I am and what I am fighting for.
If you agree, disagree, or think this is nonsense, email contact@clinicalbudget.com
If you’d like me to cover a specific topic, you can suggest it here.


Why Clinical Trial QC Feedback Often Sounds Harsher Than Intended