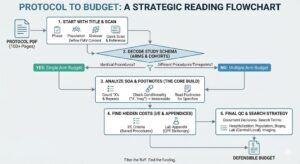
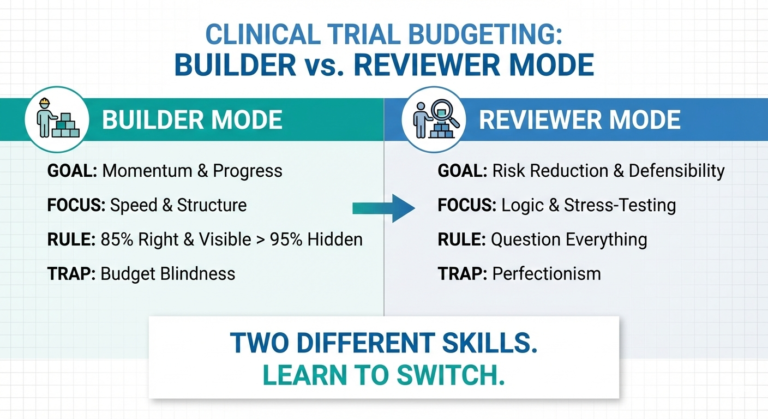
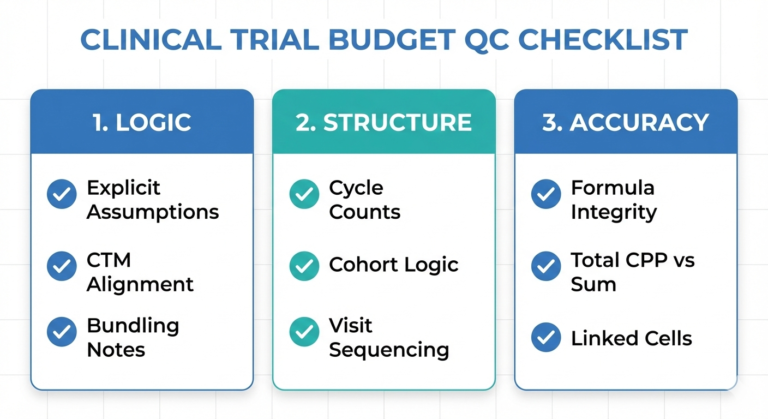
Filter out the fluff and find what actually affects your bottom line when reading a clinical trial protocol.
I don’t have a medical background and I assume you don’t either. Receiving a 100-page pdf full of jargon is always intimidating. Hell, I get the jitters when I have to do a new build even with years of experience behind me. This article focuses on how to quickly identify the protocol elements that actually affect cost, structure, and QC outcomes.
When I first started, I was told that I should read it in its entirety, pay attention and use the search function if I needed to find something specific. This was extremely unhelpful considering: a) I had no idea what I was looking for; b) we were getting pressed to have this budget finalized as soon as possible; and c) I read really, really, slowly!

Reading and understanding the protocol is an overlooked skill – but it underpins QC success, build speed, and confidence.
Study Title
At the top of your file you should see the study title. In just a few words it can tell you the study phase, the population, the disease, the type of intervention and the study design. All these factors play a role in the way Fair Market Value (FMV) costs are calculated and it can give you a hint of what you’re building for.
Table of Contents and List of Abbreviations
The Table of Contents, typically followed by the List of Abbreviations can be helpful guides throughout your building process. I recommend scanning the different section titles and making a mental note of any segment that might have bearing on any medical procedure or the study design. Do not spend a ton of time on this step, as you will naturally read most of these sections when you are going through the Schedule of Activities (SOA). Plus I actually recommend returning to this part after you think your budget is complete, just to make sure there isn’t something relevant you might have missed.
The List of Abbreviations will be your quick referencing guide for any weird amalgamation of letters you encounter. Note that in a properly built protocol all abbreviations should be included in it but things can be overlooked. If that is the case, don’t panic and ask your team or try to figure out what it means using Google.
Study Schema: Translating Design Into a Budget Structure
Time to get to the meat and potatoes! The Study Schema aims to compress complex clinical and operational information into one visual, so it can be A LOT for anyone that has not been trained to interpret it. Schemas contain a number of different elements, as it is to be used by an array of stakeholders like clinicians, Program Leads and, of course, budgeters. Our goal as builders is to figure out what information is relevant to us and what can be dismissed.
Let’s look at three common schema patterns and how to interpret them from a budgeting perspective.
Example 1 – 1-armed budget with 2 cohorts

The example above is pretty straightforward – we have 2 cohorts with identical drug administration – one being our Investigational Product (IP) and the second – a cohort that will receive a Placebo instead of the IP. In essence there is no difference between these two from a budget perspective – drug administration is the same, procedures that need to be carried out match perfectly, so unless there is a specific note in the SOA, if something is only applicable to the Placebo or the Main cohort, we can safely assume we have a single armed budget on our hands.
The distinction between arms and cohorts might be confusing in the beginning. Different cohorts can help clinicians first and foremost protect patients by identifying the optimal balance between efficacy and toxicity. Cohorts can also be used to test differences between a monotherapy vs a combination of drugs and account for biological differences and disease progression. All of these factors may or may not have bearing on the budget. It is our job to look critically at the information we are provided with, and try to determine if the differences between the cohorts warrant separating them into their own budget arms.
Example 2 – 1-armed budget with two cohorts based on patient diseases
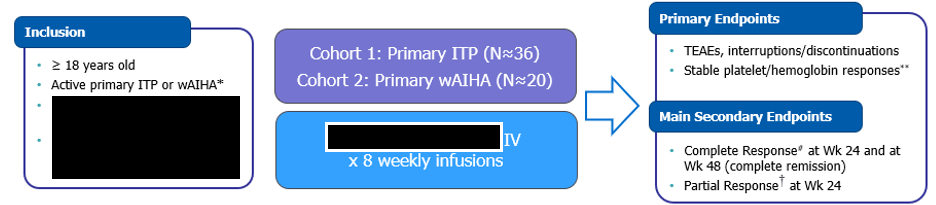
In this example, we have two diseases – Immune Thrombocytopenia (ITP) and Warm Autoimmune Hemolytic Anemia (wAIHA). While these are two very distinct cohorts, we can see that, according to the blue box, the IP administration and the study endpoints are the same. We can make a mental note that we should be seeing a lot of overlap between the two and scroll down to the SOA and check what kind of differences we would be looking at.
We should have a footnote or some kind of indication if a certain line does not apply to both cohorts, like so:
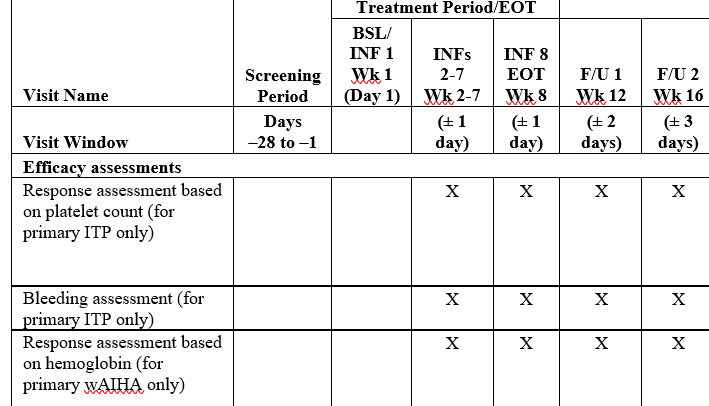
From a budget builder’s perspective, if most procedures overlap or if there is a difference but the timepoints match up, we can still have a single arm in our budget, as long as things don’t become overly complex and hard to read.
Example 3 – 2-armed budget on a multi-part study
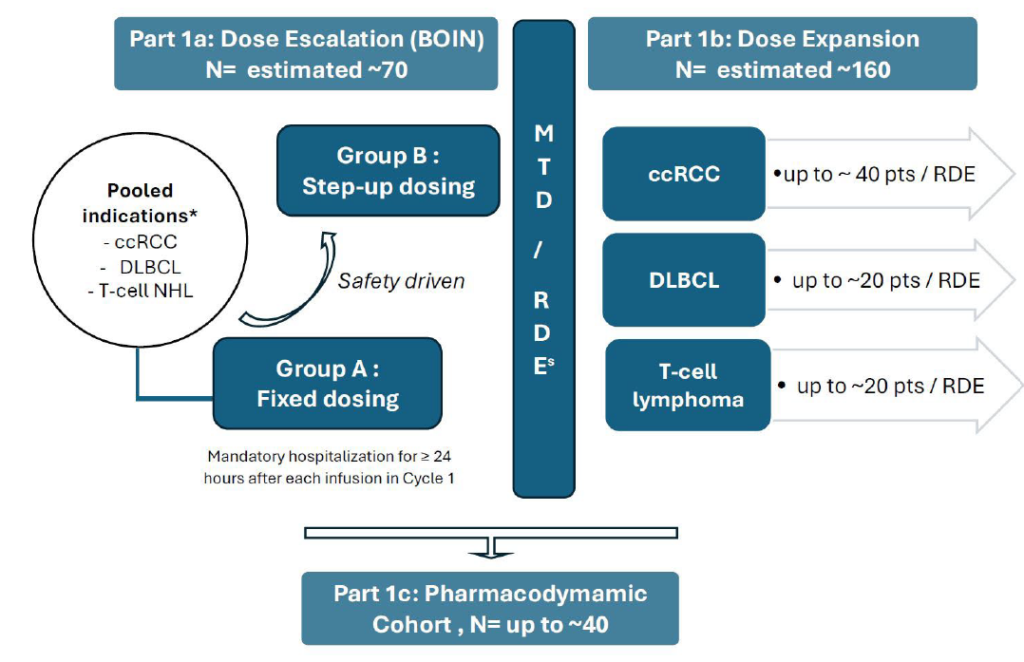
Here is where things can start to get confusing. From the get-go, I would have no clue what was going on, so I would start looking for help. There are two sections that can make a huge difference in trying to interpret a flowchart like that – the Study Design section and the SOA itself.

Here, the Study Design paragraph provides us with a couple of nuggets of information – this is a 3-part study and the budget we will be building might account for one or multiple parts of it. Participants will sequentially go through Part 1a, then Part 1b and then Part 1c. This alone isn’t enough, so I would immediately scan the SOA. I would then notice that we have only 2 SOA tables – for the benchmark group:
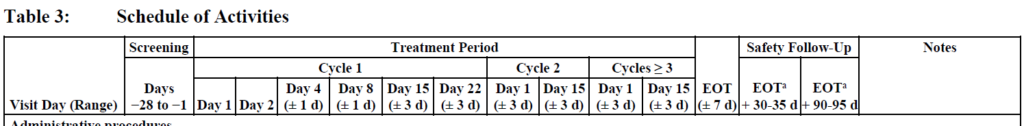
And for the step-up dosing group:

From a budgeting perspective, this schema translates to:
Two separate SOAs for Groups A and B mean that there are substantial differences between the two, so having two separate arms is warranted. Parts 1b and 1c do not seem to have any bearing on the SOA or the budgets, according to the protocol, and can be disregarded at this point.
Schedule of Activities / Schedule of Events
I doubt that I need to teach you to count the number of Xs in the SOA, but I will point out that you need to be vigilant of words that may express conditionality (“if”, “may”, “can”, “as clinically indicated”, “at investigator’s discretion”, “recommended”) or if a procedure needs to be repeated. These often end up being invoiceable procedures, particularly when frequency cannot be capped reliably, and are generally not counted towards the total cost per patient. Note that this conditionality may not have a specific timepoint tied to it, like the Coagulation panel in the example below.
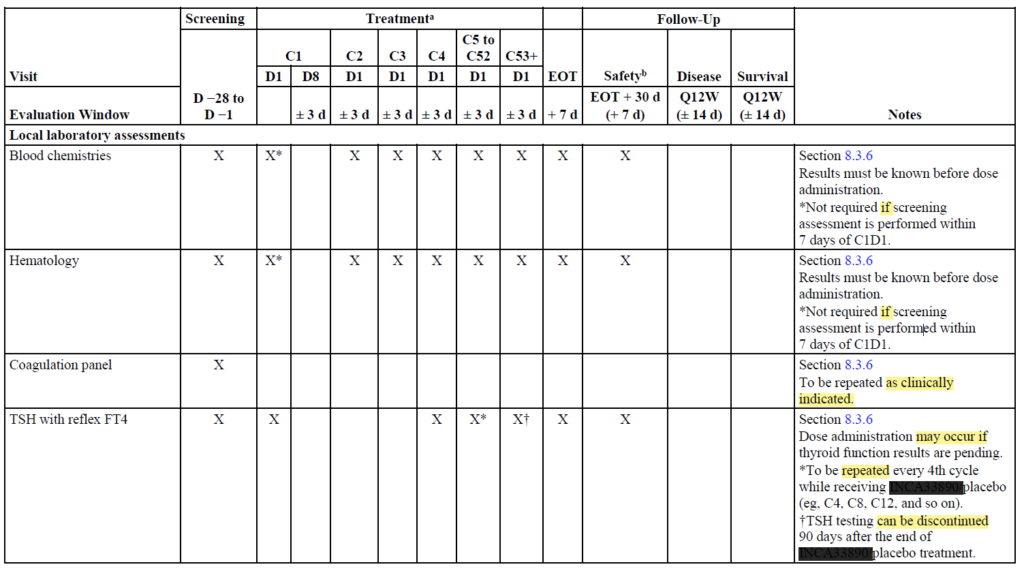
Needless to say, you should read all footnotes and relevant sections that the SOA directs you to, paying close attention to the Physical Exams/Vital Signs, ECG, Imaging Labs and PK/PD sections.
Inclusion/Exclusion Criteria and Study population
This is the segment you should go over after reviewing the SOA in its entirety. These paragraphs may hold information that is not included in the procedures table and can remain overlooked. In the example below, we can see that buried deep in the exclusion criteria, there is a scenario where an HIV RNA test may be performed but it was not mentioned in the SOA or the Labs section of the protocol:

Appendices
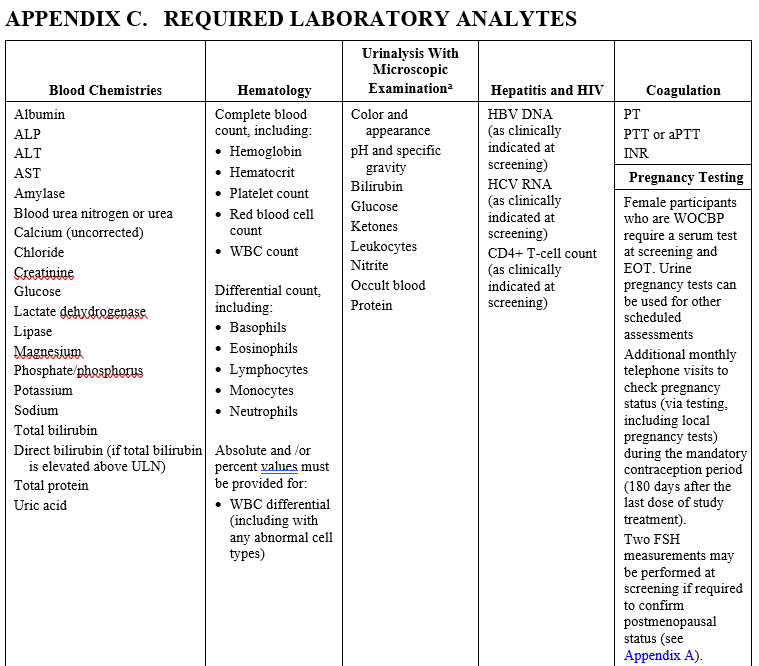
Lastly, check if there is any Appendix that is relevant to your build. For example, protocols may have a section dedicated to all the labs that will need to be performed, so use them as a guide on what precise CPT codes to use or what procedures need to be included, as invoiceable.
Tying it all together
Protocols don’t give you budgets – they give you ambiguity. Your job is to transform that ambiguity into a defensible financial plan. Now that you know the ins and outs of the entire protocol, I suggest going back to the Table of Contents and having another scan to see if something gets your attention. Consider what a reviewer might challenge and make sure that you document all your decisions, pointing to the relevant sections in the file.
Pro Tip: My Top 5 Search Terms
- Hospitalization/overnight stay
- Population
- Biopsy
- Lab (Central or Local)
- Imaging, image or simply “imag”
Even with a solid understanding of how to read a protocol, mistakes still happen. Budgeting involves judgment calls, assumptions, and trade-offs that don’t always become visible until a second set of eyes reviews the file. Many budget issues are not true “errors,” but gaps in how decisions are explained or defended during review. If you want to understand how reviewers think and why otherwise solid budgets get flagged, check out why budgets often fail QC.
Find out who I am and what I am fighting for.
If you agree, disagree, or think this is nonsense, email contact@clinicalbudget.com
If you’d like me to cover a specific topic, you can suggest it here.
Download a flowchart of how to read a protocol below ↓