If you are anything like me, you probably got into budget building on a whim rather than it being a deliberate goal you set yourself. You spent a couple of years as a budget analyst and now you are starting to wonder what the next step is. You might get the feeling that you got stuck into one track and there is no wiggle room – you will get promoted to a senior, and you will keep grinding away, while CTMs, PMs and CRAs often appear to have clearer leadership tracks and senior positions destined for them.
Nobody tells you what’s actually possible from here.
In this multi-part Series I aim to tackle this exact question. I will try to compare different roles and what they have in common with budget building, how easy would you be able to transfer, what kind of workload can you expect, and, most importantly, what kind of compensation you might be looking at.
Part 1 covers what skills you already have and offers a framework of how to think about transitioning.
Part 2 offers a framework to effectively evaluate what you should be looking for when choosing a role to transition to.
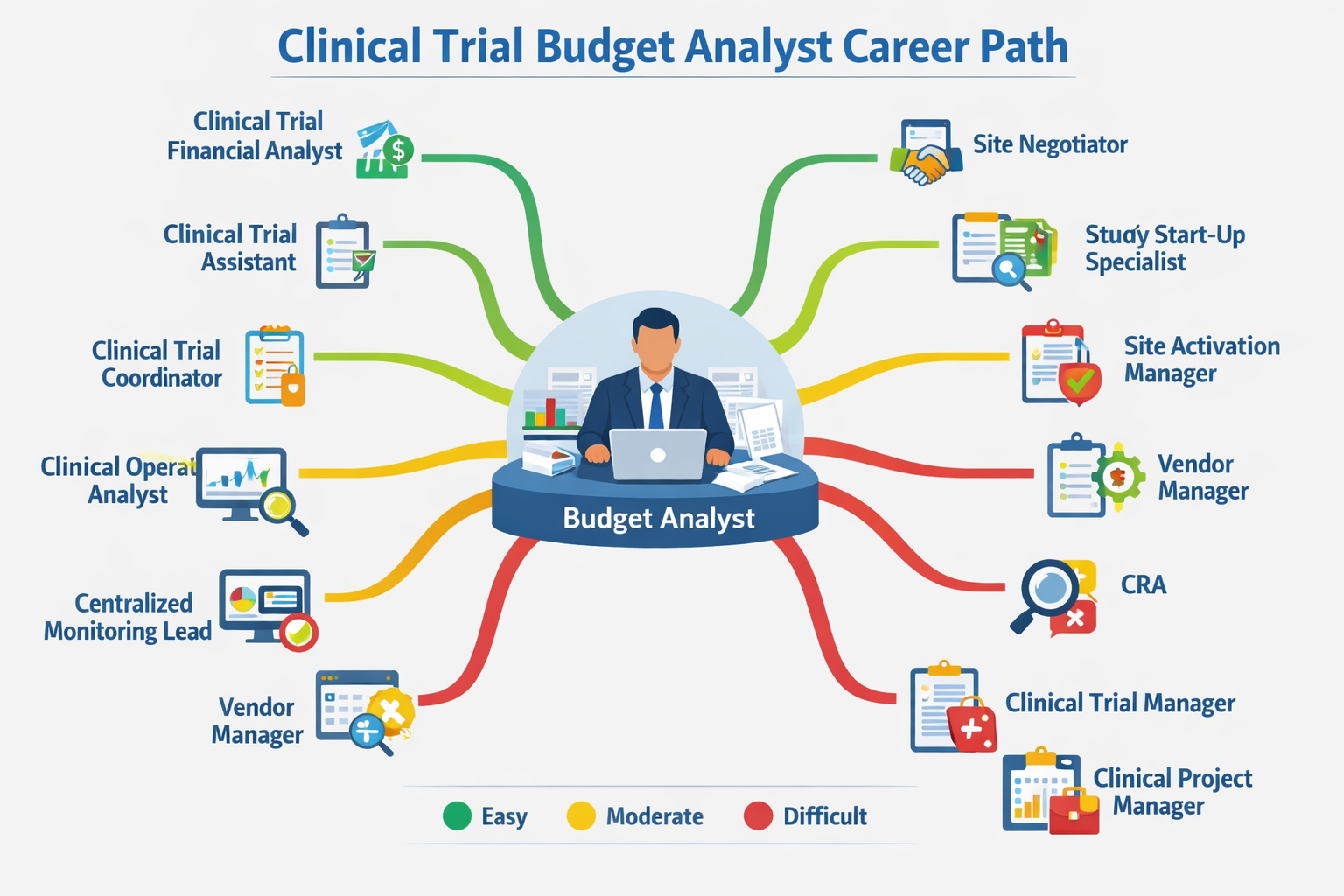
This article will focus on the actual roles you might be aiming for – I offer a table that compares different roles and what you can expect in terms of workload and salary. I also give you a short summary of each role and how you may fit into that position. It also answers the question: “What career can I get into without a science background/medical degree?”
The roles (table comparison)
Baseline Reference: Clinical Trial Budget Analyst / Budget Developer / Investigator Grants Analyst (IGA)
Before comparing where budget analysts can go, let’s take a look at where you are starting from. The table below profiles the budget analyst role using the same evaluation framework applied to all transition targets.
| Dimension | Budget Analyst / Budget Developer |
| Primary focus | Developing, negotiating, and managing clinical trial budgets; payment terms review. |
| Medical knowledge required | Low – protocol literacy required; clinical or therapeutic expertise not required |
| Skill profile | Financial analysis, contract negotiation, stakeholder communication, GCP awareness, CTMS/Excel proficiency, process improvement |
| Barrier to entry | Moderate – typically requires 2–5 years of clinical research or financial experience; bachelor’s degree is the standard |
| Workload intensity | Moderate (6-7/10) – deadline-driven during study start-up and amendment periods; more stable during study conduct |
| Salary range (US) | $62,000–$112,000 (typical); senior roles at large sponsors may reach $130,000+ |
| Career ceiling | Director of Budgets/Contracts, VP of Research Finance, Clinical Operations leadership |
| Strongest assets for transition | Budget and contract expertise; financial modelling; cross-functional stakeholder management; accountability under pressure |
The Roles: At-a-Glance Comparison
| Role | Medical Knowledge Required | Skill Overlap with IGAs | Transition Difficulty | Workload Intensity | Salary Range (US) |
| ⭐ Clinical Trial Budget Analyst/IGA (baseline) | Low | – | – | Moderate (6-7/10) | $62K–$112K |
| Clinical Trial Financial Analyst (FA) | Low | Very High | Easy | Moderate | $58K–$98K |
| Clinical Trial Assistant (CTA) | Low | High | Easy/Moderate | Moderate | $48K–$83K |
| Clinical Trial Coordinator (CTC) | Low–Moderate | Medium | Moderate | Low-Moderate (4-5/10) | $54K–$99K |
| Site Negotiator / Contract Negotiator | Low | Very High | Easy/Moderate | Moderate | $60K–$110K |
| Study Start-Up Specialist | Low | High | Moderate | Moderate/Heavy | $75K–$130K |
| Site Activation Manager (SAM) | Low | High | Moderate | Moderate–Heavy (7-8/10) | $73K–$126K |
| Clinical Operations Analyst | Low | High | Easy/Moderate | Moderate | $76K–$136K |
| Centralized Monitoring Lead (CML) | Low–Moderate | Medium | Difficult | Heavy (8-9/10) | $76K–$133K |
| Clinical Outsourcing / Vendor Manager | Low | High | Moderate | Moderate (6-8/10) | $80K–$140K |
| Clinical Research Associate (CRA) | Low–Moderate | Medium | Difficult | Heavy (8-9/10) | $87K–$148K |
| Clinical Trial Manager (CTM) | Low–Moderate | High | Moderate/Difficult | Heavy (8-10/10) | $104K–$162K |
| Clinical Project Manager / Project Lead | Low–Moderate | High | Difficult | Heavy (8-10/10) | $91K–$141K |
A few notes on how to read this table:
- Salary ranges are based on US market data and span entry to senior-level positions within each role. While these ranges are very highly dependent on the region and the company, they can serve as a baseline for comparison between the various roles.
- Medical knowledge ratings reflect what is genuinely required to perform the job – not what might appear in a preferred qualifications section.
- Barrier to entry reflects the realistic difficulty of making the transition from a budget analyst background specifically, not from clinical research generally.
1. Can a Budget Analyst become a Clinical Trial Financial Analyst (FA)
Where a budget analyst focuses on the creation and negotiation of site-level costs, the Clinical Trial Financial Analyst focuses on the full financial lifecycle of a study: tracking actuals against forecasts, producing financial reports for study teams and leadership, managing revenue recognition, and ensuring that the sponsor’s investment is being deployed efficiently across the trial.
The day-to-day work involves monitoring study budgets in real time, reconciling investigator payments against contracted amounts, forecasting burn rates at the site and study level, and flagging variances before they become problems. At larger sponsors and CROs, the role also involves building and maintaining financial models that project total study cost across different enrollment scenarios – the kind of modeling that becomes critical when a sponsor needs to decide whether to extend a study, add sites, or cut scope.
- Why it fits: You already understand pass-through costs, investigator payments, and Fair Market Value (FMV).
- Direct Skills Transfer: Budget forecasting, variance analysis, and monthly revenue cycles.
- Key Growth Area: Learning project-based accounting and revenue recognition principles (e.g., SAP or Oracle EPM); financial modeling expertise.
2. Can a Budget Analyst become a Clinical Trial Assistant (CTA)
The CTA supports the preparation, review, and tracking of clinical trial agreements, including Clinical Trial Agreements, Site Agreements, and Confidentiality Agreements. The role involves maintaining study files, tracking study progress, preparing trial documents, and providing administrative and operational support to clinical research teams, with a strong emphasis on organization, attention to detail, and communication.
For budget analysts seeking a lower-risk lateral move – perhaps one that preserves their current skill set while opening new career pathways – a contracts or CTA role is a logical bridge. It is less of a career leap and more of a repositioning. The lower salary ceiling is the main trade-off.
- Why it fits: The CTA role is the administrative backbone of clinical operations. A budget analyst’s familiarity with the documentation required for site payments and financial tracking ensures that the Trial Master File (TMF) remains accurate and audit-ready.
- Direct Skills Transfer: Document version control, tracking study milestones, and maintaining organizational spreadsheets.
- Key Growth Area: Deepening knowledge of Good Clinical Practice (GCP) and non-financial essential documents (e.g., 1572s, CVs, and medical licenses).
3. Can a Budget Analyst become a Clinical Trial Coordinator (CTC)
A more “generalist” administrative entry level role that supports the PM and CRA. The CTC is primarily a site-level position – managing the day-to-day operations of clinical trials at the investigator site, including participant recruitment, protocol compliance, data entry, and regulatory document management For budget analysts, the skill overlap is medium rather than high: the organizational, documentation, and protocol-literacy skills transfer, however, the patient-facing and clinical coordination aspects of the role require exposure that most budget analysts do not have. The CTC role is best understood as a lateral move that widens operational experience – useful as a bridge, but not an obvious upgrade from a financial perspective.
- Why it fits: While the budget analyst looks at the “what it costs,” the CTC looks at “how it happens.” The analyst’s ability to take responsibility for a decision – such as why a specific site was granted a certain overhead – translates well to managing site-level logistics.
- Direct Skills Transfer: Stakeholder communication, visit scheduling logic, and site-level accountability.
- Key Growth Area: Patient recruitment strategies and understanding the “clinical” side of patient safety reporting.
4. Can a Budget Analyst become a Site/Contract Negotiator
The Site Negotiator sits at the intersection of contracts and budgets, responsible for reaching executable agreements with investigative sites on both the financial terms and the legal language governing a trial. In practice this means running parallel negotiations — often across dozens of sites simultaneously — managing the back-and-forth on payment schedules, overhead rates, and contract clauses until both parties reach a position they can sign.
Where a budget analyst builds the financial case and a contracts specialist manages the documentation, the site negotiator owns the conversation from opening position to signature. That requires a specific combination of commercial confidence, patience, and the ability to read when a site is genuinely stuck versus when they are testing your flexibility.
For analysts who want to move laterally into a role that explicitly names their existing skills as the core competency, this is it.
- Why it fits: This is the most direct lateral move. Negotiators often work side-by-side with budget analysts. The ability to argue for a specific position and defend it against site pushback is the primary requirement here.
- Direct Skills Transfer: Master Service Agreement (MSA) familiarity, Fair Market Value (FMV) benchmarking, and redlining legal/financial terms.
- Key Growth Area: Legal frameworks including indemnification, intellectual property (IP) rights, and subject injury clauses.
5. Can a Budget Analyst become a Study Start-Up / Site Activation Specialist
Study Start-Up/Site Activation Specialists manage the critical path from a signed protocol to a site that is ready to enroll patients. Study start-up (SSU) is one of the most complex and heavily regulated parts of any clinical trial or program launch, involving a significant number of stakeholders at start-up sites, regulatory agencies, and vendors. Practically speaking, the day-to-day involves coordinating regulatory and ethics submissions, negotiating site contracts and budgets, collecting and reviewing essential documents, and tracking activation milestones across multiple sites simultaneously.
This is one of the most natural lateral transitions available to budget analysts. The core of SSU work – negotiating site budgets, reviewing investigator contracts, tracking deliverables across multiple sites – is work that budget analysts already do, just framed differently. The budget negotiation component is effectively the same function: working with sponsors and sites to agree on fair and compliant financial terms before the study begins. Budget analysts who have worked on study start-up budgets will recognize the majority of the SSU workflow.
- Why it fits: Site activation is the stage where trials most often stall due to budgeting delays. An analyst who knows how to unblock a financial negotiation can accelerate the “green light” process significantly.
- Direct Skills Transfer: Protocol analysis for “Schedule of Assessments” (SoA) and critical path management.
- Key Growth Area: Local and central Institutional Review Board (IRB) or Ethics Committee (EC) submission processes.
6. Can a Budget Analyst become a Site Activation Manager (SAM)
The Site Activation Manager sits one level above the Site Activation Specialist and carries full ownership of the site start-up function across a study or portfolio of studies. This includes developing and maintaining the Site Activation Management Plan, managing cross-functional teams, tracking KPIs, and ensuring that sites are activated on time and in compliance with GCP, ICH, and sponsor requirements.
The SAM role combines two things budget analysts already do well – budget and contract management – with a third thing they may need to develop: operational team leadership and site-level delivery oversight. A budget analyst who moves first into a Site Activation Specialist or Study Start-Up role and builds two to three years of direct activation experience would have a comparable and arguably more direct route to the SAM level.
- Why it fits: This is a leadership role. A budget analyst’s experience in taking responsibility for the financial health of a project prepares them to own the activation timelines for an entire region or country. Budgeting is the #1 delay in site activation, a SAM with a budgeting background is a “hired gun” who knows how to fix the most common problem.
- Direct Skills Transfer: Risk mitigation and high-level project forecasting.
- Key Growth Area: Cross-functional leadership and managing team-wide KPIs rather than individual tasks.
7. Can a Budget Analyst become a Clinical Operations Analyst
The Clinical Operations Analyst is a data and process-oriented role that supports clinical trial execution by tracking operational metrics, identifying performance trends, managing CTMS data, and producing reports that help study teams and leadership make better decisions. This position combines data analysis, day-to-day trial operations, and technology – requiring comfort with systems, structured data, and analytical thinking alongside an understanding of how clinical trials run.
This role has grown in prominence as sponsors and CROs have invested more heavily in real-time operational intelligence. The ability to turn raw CTMS data into actionable insights – enrollment forecasts, site performance scorecards, budget vs. actual comparisons – is increasingly valued across the industry.
The transition is natural because the underlying logic is the same. A budget analyst who tracks variances across a portfolio of studies, flags cost outliers, and produces financial reports for leadership is already performing a version of these duties. The difference is the data source – operational metrics rather than financial ones – and the audience, which shifts from finance toward clinical operations and project management.
- Why it fits: You should already be proficient in Excel and you might have working knowledge of Power BI. This role takes those technical skills and applies them to study metrics (KRIs).
- Direct Skills Transfer: Trend analysis and data-driven forecasting.
- Key Growth Area: Developing Key Risk Indicators (KRIs) and statistical software. Analysts with exposure to tools like Medidata Rave, Veeva Vault, or Oracle Clinical will have a meaningful advantage.
8. Can a Budget Analyst become a Centralized Monitoring Lead (CML)
The Centralized Monitoring Lead is a relatively recent but fast-growing role in clinical research, born directly out of the shift towards risk-based monitoring. Rather than conducting traditional on-site monitoring visits, the CML oversees the trial remotely – using data analytics, CTMS platforms, and electronic data capture systems to identify risks, flag trends, and coordinate corrective actions across sites without ever needing to set foot in a clinic.
The role sits at an intersection that is increasingly valued by sponsors and CROs: it combines the clinical oversight sensibility of a monitor with the data fluency of an analyst – and it does so without the heavy travel burden that traditional CRA work carries.
The CML requires critical thinking and analytical skills to understand and analyze complex clinical data, provide insight into risk reports, identify trends and outliers, and has a strong preference for therapeutic area-specific experience and knowledge. The clinical data review component – reviewing patient-level data for safety signals, protocol deviations, and data integrity issues – is territory that budget analysts have not typically operated in. You may want to consider exploring the Centralized Monitoring Assistant (CMA) role, which has a considerably lower barrier of entry.
- Why it fits: You understand the operational fallout of a poorly built budget or database. In this role, you use that foresight to monitor study alerts and compliance.
- Direct Skills Transfer: Risk assessment and mitigation planning.
- Key Growth Area: Deepening your GCP (Good Clinical Practice) knowledge and remote monitoring technologies.
9. Can a Budget Analyst become a Outsourcing & Vendor Manager
Clinical outsourcing and vendor management roles sit at the intersection of procurement, contract management, and clinical operations. This role is heavily focused on coordination and negotiation and requires a strong command of how CRO and vendor contracts are structured, how budgets are built and monitored, and how performance is measured and enforced across the study lifecycle.
Responsibilities in clinical outsourcing roles explicitly include assisting with clinical trial budget modeling, creating what-if scenarios for future trials, and setting up cost-saving tracking tools – tasks that budget analysts perform as a matter of routine. The transition asks for an expansion of scope, not a pivot in direction.
- Why it fits: You are an expert at identifying “scope creep” – where a vendor’s activities exceed the budget.
- Direct Skills Transfer: Negotiating scope and cost changes, budget and contract knowledge.
- Key Growth Area: Strategic relationship management and understanding global outsourcing strategies.
10. Can a Budget Analyst become a Clinical Research Associate (CRA) / In-House CRA (IHCR)
The CRA role is the most common path in clinical research – and the most frequently misunderstood as a natural next step for budget analysts. CRAs ensure that trials at investigator sites comply with the protocol, regulatory requirements, and GCP standards, with typical responsibilities including auditing patient records, verifying informed consent, overseeing site operations, and ensuring data accuracy.
CRA work is site-facing, monitoring-focused, and heavily travel-dependent (unless you are ready to sacrifice some of your salary for an in-house CRA role). Budget analysts without site-level experience will typically need to bridge through a CTA or study coordinator role first. The workload and travel intensity are also among the highest on this list – a meaningful lifestyle consideration.
- Why it fits: Budget analysts understand the frequency and intensity of study visits because they have priced them. This helps a CRA understand why certain data points are “critical” from a financial and compliance perspective.
- Direct Skills Transfer: Data verification and meticulous attention to protocol adherence.
- Key Growth Area: On-site monitoring techniques, source data verification (SDV), and GCP knowledge. You need to bridge the gap between “looking at a protocol for costs” and “looking at a protocol for patient safety.”
11. Can a Budget Analyst become a Clinical Trial Manager (CTM)
The CTM is the operational lead for a clinical trial, responsible for everything from study planning and site activation through to close-out. Once a trial is in motion, the CTM’s role centers on oversight and coordination – conducting regular meetings to monitor timelines, identify potential risks, and track milestones, while collaborating cross-functionally to ensure all teams, from data management to safety, are aligned and working toward the same goals.
Budget analysts already work closely with CTMs, review the same protocols, manage the same budgets, and navigate the same sponsor relationships. The key difference is accountability scope: a CTM owns the entire operational delivery of a study, not just its financial picture. Budget analysts who have been embedded in study teams – attending team calls, contributing to sponsor meetings, reviewing protocol amendments – will find the transition more natural than those who have operated in a more siloed finance function.
The education and experience requirements for a CTM can vary by employer, but they generally need several years of experience in clinical research – often having worked in roles such as clinical research associate, project manager, or clinical data manager before advancing to the CTM level. Medical knowledge in the clinical sense – understanding treatment mechanisms, patient safety signals, or scientific methodology at depth – is typically not what CTM job descriptions are testing for. What is non-negotiable is GCP knowledge and operational experience.
- Why it fits: Most CTMs struggle with the financial side of their studies. A CTM who was previously a Budget Analyst brings budget knowledge and understanding to the role looks at overspending and can identify unreasonable site costs.
- Direct Skills Transfer: Budget oversight, vendor management, stakeholder negotiation, GCP awareness, and the ability to defend decisions under pressure.
- Key Growth Area: Therapeutic area expertise (e.g., Oncology, Cardiology) and managing global clinical teams.
12. Can a Budget Analyst become a Clinical Project Manager / Project Lead (CPM / PM / PL)
PMs play a vital role, overseeing the successful execution of clinical trials from inception to completion. In practical terms, being a CPM means being responsible for setting up, coordinating, and leading clinical studies – often aided by an Assistant CPM – while leading a cross-functional clinical project team that includes data managers, monitors, and vendors such as central laboratories, and providing regular progress updates to sponsors or clients. The CPM owns the study from end to end: timelines, budget, quality, team performance, vendor relationships, and regulatory compliance all fall within their remit simultaneously.
CPMs (should) bring a unique blend of skills to the table: strong attention to detail, the ability to track multiple deadlines and budgets, team leadership, collaboration and organizational abilities, excellent communication and interpersonal skills, and problem-solving capability for navigating challenges that arise during the trial. On paper, this reads like a description of what a senior budget analyst already does. Where CPMs often need to build financial competency, budget analysts already have it. What they typically lack is the operational and team leadership experience that rounds out the PM profile.
- Why it fits: The PM is ultimately accountable for the study’s profitability and success. A background in budgeting provides a “Financial Anchor,” allowing the PM to make decisions that protect the sponsor’s investment while ensuring trial integrity.
- Direct Skills Transfer: High-stakes stakeholder management and the ability to argue for project resources.
- Key Growth Area: The overlap is real but the distance is significant. Strategic planning, client relationship management, and managing the “Triple Constraint” (Time, Cost, Scope). For those aiming for a CPM role that allows for growth, obtaining the ACRP-PM certification is considered the ideal approach. The PMP certification from PMI is equally well-regarded, particularly at sponsor-side organisations and in roles where project management methodology is explicitly valued.
Key Insight: The “Hybrid” Advantage
The clinical research industry is in the middle of a structural shift. The era of the pure specialist – the monitor who only monitors, the budget analyst who only builds budgets – is giving way to something more valuable: the hybrid professional who can move fluidly between financial, operational, and strategic functions.
This matters for budget analysts specifically because you are already further along that path than you probably realize. You have financial literacy that most operational roles lack; you possess protocol familiarity that most finance specialists do not; you can rely on negotiation experience that most analytical roles find wanting. The question is not whether you have the foundation – it is which direction you want to build on top of it.
The roles in this article are not a ranked list of difficulty. They are a map of adjacent territory. Some of that territory is a short walk from where you are; some requires building new muscle first.
You can get a quick infographic of the post popular roles you can transition to and difficult it would be below:
Find out who I am and what I am fighting for.
If you agree, disagree, or think this is nonsense, email contact@clinicalbudget.com
If you’d like me to cover a specific topic, you can suggest it here.




Why Clinical Trial QC Feedback Often Sounds Harsher Than Intended