Budget analysts often get stuck in one track
If you are anything like me, you probably got into budget building on a whim rather than it being a deliberate goal you set yourself. You spent a couple of years as a budget analyst and now you are starting to wonder what the next step is and what your career path might look like. You get the feeling that you got stuck into one track with little to no option for a career transition – you will get promoted to a senior, and you will keep grinding away, while CTMs, PMs and CRAs often appear to have clearer leadership and senior positions destined for them.
Nobody tells you what’s actually possible from here.
In this multi-part series I aim to tackle this exact question. I will try to compare different roles and what they have in common with budget building, how easy you would be able to transfer, what kind of workload can you expect, and, most importantly, what kind of compensation you might be looking at.
This article will cover what skills you already have and offer a framework of how to think about transitioning in clinical research.
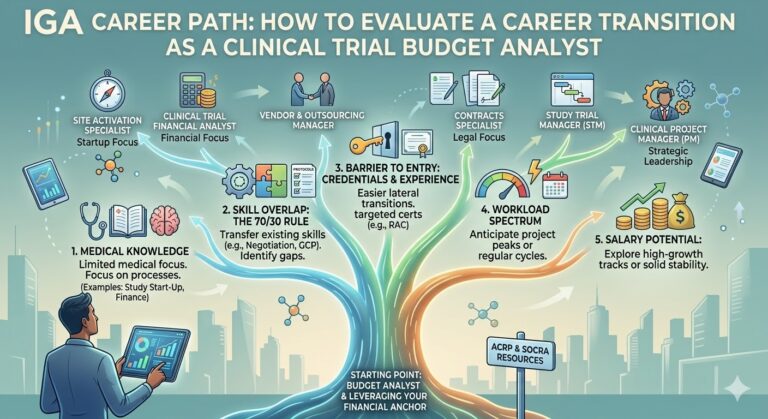
Part 2 will provide you with a framework to effectively evaluate what you should be looking for when choosing a role to transition to.
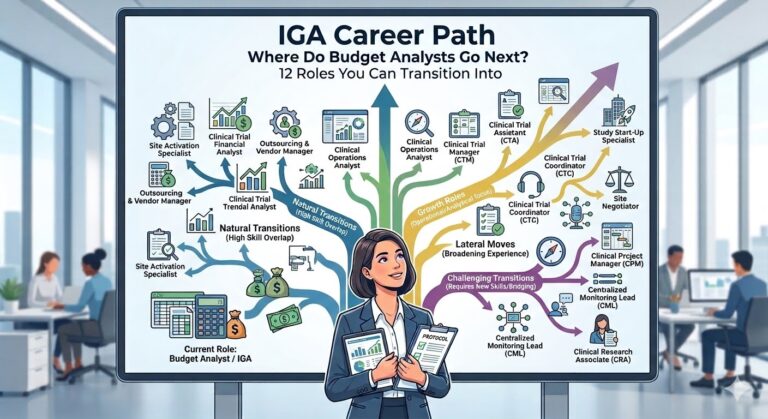
Part 3 will focus on roles in clinical research without a medical degree that you might be aiming for. I offer a table that compares different clinical trial career paths and what you can expect in terms of workload and salary. I also give you a short summary of each role and how you my may fit into that role.
What skills budget analysts already have
Before looking outward at new job titles, it is essential to deconstruct the “Budget Analyst” role into its core competencies. A Clinical Trial Budget Analyst is responsible for developing, negotiating, and managing budgets for clinical trials, working closely with research teams, sponsors/CROs, and site payments to ensure that trial costs are accurately estimated and allocated. Their role includes analyzing financial contracts, monitoring costs, and ensuring compliance with regulatory and organizational guidelines. Below, I go over several distinct professional competencies, each transferable.
1. Advanced Protocol Deconstruction
A budget analyst’s most valuable “clinical” skill is the ability to perform a line-item deconstruction of a Clinical Trial Protocol. While a CRA looks at a protocol for monitoring compliance, a budget analyst translates the Schedule of Assessments (SoA) into a financial and operational roadmap.
To build a budget, you first have to understand what is being budgeted (duh). This means budget analysts routinely read protocols, understand study visit structures and are aware of standard of care and research-specific procedures. That knowledge is exactly what clinical operations professionals work with every day. The gap between a budget analyst and a Study Coordinator or Clinical Trial Manager, in terms of protocol familiarity, is smaller than it looks on paper.
- Transferable Value: This deep understanding of study visits, procedures, and “Standard of Care” versus “Study Related” costs is exactly what is required for Study Start-Up (SSU) and Clinical Project Management.
2.Negotiation and Conflict Resolution
Budgeting is never a passive activity. Analysts spend a significant portion of their time negotiating with investigative sites, SMOs, CROs and sponsors. They must find a balance between the sponsor’s financial constraints and the site’s willingness to join the trial and internal pricing lists. This requires sophisticated communication skills which are directly relevant to roles in Contracts and Agreements, Vendor Management, and Clinical Project Management, where setting expectations and coordinating across stakeholders is central to the job.
- Transferable Value: These are high-stakes soft skills. The ability to defend a position using data – while maintaining a professional relationship – is the hallmark of an effective Vendor Manager or Contracts Specialist.
3. Project Management Fundamentals
Budget analysts must coordinate budget negotiations, track frequent protocol amendments, and manage multi-site trial costs, while collaborating with project managers, negotiators, and site payments teams to ensure all parties are aligned on budgets, timelines and compliance. Managing a portfolio of studies at different stages of negotiation – across multiple sponsors, sites, and therapeutic areas – Clinical trial budgeting is project management in everything but name. Many IGAs are already doing the substantive work of a project coordinator; they simply lack the formal title.
- Transferable Value: Managing a portfolio of budgets across multiple therapeutic areas and phases is identical to the work of a Project Coordinator or Study Start-Up Manager. The ability to maintain timelines while balancing the needs of disparate stakeholders is the core requirement for any operational leadership role.
4. Process Improvement Orientation
In a growth environment, budget analysts are expected to identify opportunities for process improvement within the contracts and budgets team and contribute to the development and implementation of best practices, as well as provide training and support to team members with budget-related questions. Noticing patterns that stall negotiations, missing language in the payment terms or identifying country specifics that others need to be trained on translate into quality, operations, and management roles alike.
- Transferable Value: This orientation is highly prized in Quality Assurance (QA), Clinical Operations Management, and Corporate Strategy. The ability to train others and develop standardized workflows proves that you can scale operations, a key requirement for moving into Director-level positions.
5. Data Integrity and Financial Forecasting
A minor error in a per-patient grant can result in millions of dollars in variances across a global Phase III trial. That is why both Builders and Reviewers can hone on the tiniest discrepancy and ask the right questions.
- Transferable Value: These skills are highly sought after in Clinical Operations Analytics and Data Management, where the ability to spot trends early and outliers within large datasets is critical.
6. Financial and data systems proficiency
Strong budget analysts demonstrate advanced Excel skills and competence in spreadsheet and database software. Familiarity with CTMS platforms, financial planning tools, and structured data workflows is a genuine differentiator in an industry that is increasingly technology-driven. This technical fluency translates well into Operations Analyst, Data Management, and Clinical Systems roles.
- Transferable Value: If you can manage a Clinical Trial Management System (CTMS) and structured data workflows for global budgets, you can also handle overseeing the flow of clinical data.
7. Limited Regulatory and Compliance Literacy
The regulatory landscape regarding Fair Market Value (FMV) and transparency (such as the Sunshine Act) is more stringent than ever. Budget analysts are often the primary gatekeepers of financial compliance, ensuring that payments to healthcare professionals do not violate anti-kickback or global anti-corruption laws.
While budget analysts are not regulatory specialists, they operate in a compliance-adjacent space that gives them a meaningful head start when moving into roles that require GCP knowledge – which, in practice, is most clinical research roles.
- Transferable Value: This expertise creates a natural bridge into Regulatory Affairs or Quality & GCP Auditing, where financial compliance and clinical integrity intersect.
8. The Invisible Asset: Accountability and the ability to defend your decisions
Another that rarely appears in a job description but shows up constantly in practice: the ability to own a decision, stand behind it, and articulate clearly why it was made. Clinical trial budget analysts develop this capacity through necessity. Every budget is a series of judgment calls – which costs are billable and which are invoiceable, where to push back on site requests – and those calls can be challenged at any point by a sponsor, a finance director, a site, or an auditor. Analysts who last in this role learn not just how to make defensible decisions, but how to walk someone through the reasoning behind them under pressure.
This is not the same as stubbornness. It is the combination of analytical confidence and professional composure – knowing your numbers, knowing your rationale, and being willing to hold the line when you are right while remaining genuinely open to revision when new information warrants it. In clinical research, where ambiguity is constant and stakeholders often have competing priorities, that disposition is rare and valuable. It transfers directly into roles like Clinical Trial Manager, Contracts Specialist, and Vendor Manager, where you will regularly be asked to justify timelines, defend contract positions, or explain cost overruns to people who are not happy to hear the answer.
Accountability in this role means not just “doing the math,” but standing behind the results if a trial goes over budget or if a site questions the Fair Market Value (FMV) benchmarks. If a budget analyst realizes an error was made in a global template, the “accountable” move is to flag it immediately, propose the fix, and explain the impact.
- The Transferable Value: This builds executive presence. Senior leaders (Directors and VPs) don’t look for people who never make mistakes; they look for people who own their decisions, navigate the fallout with transparency, and provide solutions. This is particularly vital in Centralized Monitoring, where you must defend why you flagged (or didn’t flag) a specific site risk. In roles like Project Management, Study Start up or Vendor Management, you are no longer just responsible for your own tasks; you are responsible for the project’s success.
Bottom line
Taken together, this is a strong and genuinely transferable skill set. The challenge for most budget analysts is not capability – it is visibility. They often underestimate how much operational and clinical knowledge they have accumulated, and how directly it maps onto adjacent roles. Consider the big picture and reframe day-to-day task coordination as project management, query resolution as data quality and risk management, and vendor oversight as stakeholder coordination – the same principle applies to budget work. A negotiated site budget in essence is also a contract; a portfolio of study budgets is also a project pipeline.
Find out who I am and what I am fighting for.
If you agree, disagree, or think this is nonsense, email contact@clinicalbudget.com
If you’d like me to cover a specific topic, you can suggest it here.





Why Clinical Trial QC Feedback Often Sounds Harsher Than Intended