Clinical Trial Budget Bidding 101: Pre-award Budgets (Bids), Assumptions & CPT Codes

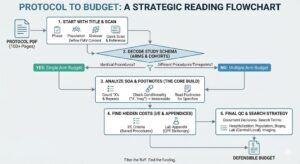
Clinical trial budget bidding begins long before a protocol is finalized. Budget builders must translate incomplete information into realistic cost estimates while balancing assumptions, CPT codes, and sponsor expectations. Understanding how to structure a pre-award budget is essential for producing bids that are both defensible and operationally workable.