Why Clinical Trial Budgets Fail QC (and Five Steps to Avoid It)
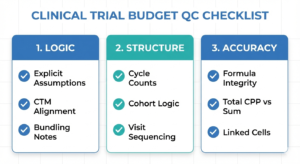
Clinical trial budgets rarely fail QC because of one big mistake. They fail because of small, compounding issues—unclear assumptions, weak structure, or broken formulas—that signal risk to a reviewer.
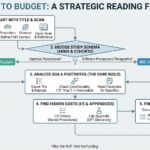
QC is about defensibility, not perfection. This article explains the most common budget QC failure patterns and five steps to reduce rework and approval delays.