Most budget analysts don’t plan their way into the role. A few years in, the question becomes: what’s next? If that question comes with a low-grade sense that you’re falling behind or missing something – that feeling has a structural explanation – and it’s worth separating it from the career question itself.
Nobody tells you what’s actually possible from here.
In this multi-part series I aim to tackle this exact question. I will try to compare different roles and what they have in common with budget building, what a clinical trial career transition looks like, what kind of workload can you expect, and, most importantly, what kind of compensation you might be looking at.
Part 1 covers what skills you already have and offers a framework of how to think about transitioning.
This article provides you with a clear framework to effectively evaluate what you should be looking for when choosing a role to transition to.
Part 3 will focus on the actual roles you might be aiming for – I offer a table that compares different roles and what you can expect in terms of workload and salary. I also give you a short summary of each role and how you may fit into that position.
How to evaluate clinical trial career transitions
Not all career moves are created equal. Some pivots from clinical trial budgeting are genuinely straightforward – a lateral shift where most of your skills transfer immediately and the learning curve is manageable. Others require real investment: new certifications, new operational experience, or time spent building credibility in a different part of the organization. Knowing the difference upfront saves you from pursuing a role that looks appealing at first glance, but proves harder to land than expected.
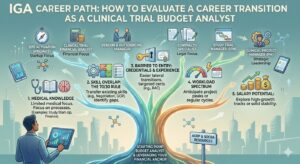
Transitioning from a specialized niche like budgeting into an operational or leadership role requires a clear plan. Using my Role Evaluation Framework, budget analysts can weigh their next move based on five critical factors – medical knowledge requirements, skills overlap, barrier to entry, workload intensity and last but not least – salary potential.
One practical point – typically transitions are easier internally, so trying to make a switch while also changing companies might make things harder, so plan accordingly.
How much medical knowledge do you need to change roles in clinical research
This is the single most important filter for budget analysts considering a career transition. You might be surprised to find that many adjacent roles that you already work with require little to no medical background – they prioritize operational competence, regulatory awareness, and stakeholder management instead. Others, particularly those closer to site-level clinical operations or medical affairs, may expect therapeutic area expertise or a science degree.
A personal friend who transitioned into a Study Trial Manager (STM) role after a career path through in-house CRA and Project Management Assistant positions noted that medical knowledge was not a core requirement of their STM work – though they acknowledged that expectations can vary from company to company. This reflects an industry pattern – the degree to which clinical research roles require medical knowledge depends heavily on the organization, the sponsor and the specific study portfolio, not just the role title.
Budget analysts often worry that their lack of a medical degree will limit their progression. However, many “high-impact” roles focus on process, efficiency and data rather than clinical diagnosis.
- The Strategy: Focus on roles where the “Low to Moderate” medical knowledge requirement is the standard. For instance, a Study Start-Up Manager needs to understand the process of a trial more than the mechanism of action of the drug.
Examples of roles typically requiring limited medical knowledge include:
- Site Activation Specialists
- Clinical Trial Financial Analysts
- Vendor and Outsourcing Managers
- Contracts Specialists
In contrast, positions in Biostatistics or Pharmacovigilance may require specialized scientific or statistical training.
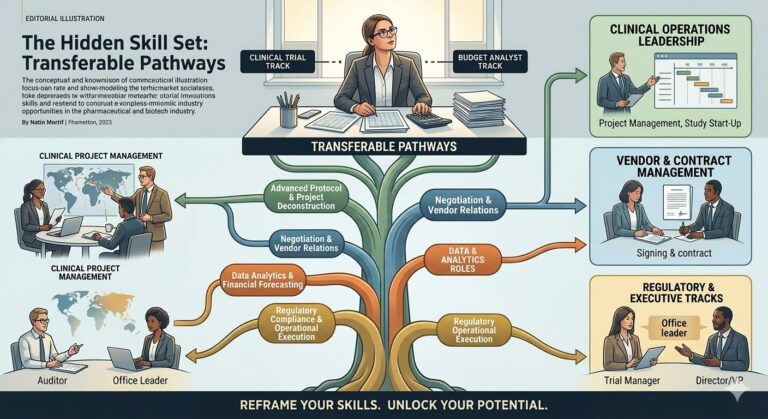
Skill Overlap With Your Current Role
As a start, focus on job descriptions. Pull two or three live postings for the role you are targeting from companies like IQVIA, Parexel, ICON, or Thermo Fisher/PPD, and compare their required competencies against what you do today. Feed them into an LLM and discuss the tasks that are expected of that role and if the skills required overlap with your current responsibilities. Does the role you aim for require protocol review? Budget or financial analysis? Stakeholder negotiation? CTMS proficiency? GCP knowledge? When updating your CV, expressing your skills in the language that companies use in job descriptions will help you get through the HR filter and move forward in the selection process.
Map the role against what you already do, identify what’s missing, and make a plan to fill those gaps. If you have not thought explicitly about what your current skill set actually contains, the builder vs. reviewer distinction is a useful place to start – most budget analysts are running two separate cognitive modes without realizing it, and both travel well. Some roles overlap heavily with budgeting skills, while others require building entirely new competencies. The most successful transitions occur when you can use 70% of your existing skills while learning 30% new ones.
High-overlap roles allow you to remain the “subject matter expert” while expanding your scope and may include:
- Clinical Trial Financial Analyst
- Vendor Management
- Contracts and Outsourcing roles
- Site Activation
Lower-overlap roles may include:
- Clinical Project Manager – While your budgeting background is a strong asset, budget analysts are not directly responsible for strategic planning, client relationship management, and managing the “Triple Constraint” (Time, Cost, Scope).
- Centralized Monitoring Lead – Budget analysts look for financial or legal outliers, CMLs look for clinical data outliers where Risk-Based Quality Management (RBQM) and data trends related to patient safety and primary endpoints are needed.
- Clinical Trial Coordinator – Going from Budget to CTC means moving from a high-level strategic/financial macro role into a high-intensity, administrative, patient-facing micro role.
Realistic Barrier to Entry
Some roles are accessible with a strong resume and a targeted cover letter. Others have hard entry requirements – years of monitoring experience, specific software certifications, or prior clinical site work, that cannot be easily reframed or substituted. When considering a career switch in clinical research, it is important to identify where you are in your own readiness and to recognize that some roles have requirements that function as genuine barriers, not just preferences.
For budget analysts specifically, the most accessible transitions tend to be roles where the primary value is operational and financial acumen – contracts management, study start-up, clinical operations analysis. The harder transitions are those where the primary value comes from field monitoring experience or therapeutic area depth, because those are things you cannot easily acquire without time in the field.
Evaluate the “credentialing” required. Some paths, like Regulatory Affairs, may require specific certifications (e.g., RAC). Others, like Site Activation, are often accessible through internal lateral moves within a CRO.
Lower barriers
- Site Activation Specialist
- Vendor Manager
- Clinical Finance roles
Higher barriers
- Regulatory Affairs Specialist
- Biostatistician
- Pharmacovigilance Scientist
Workload Intensity
Clinical research is a high-pressure environment across the board, but the shape of that pressure varies significantly by role. It helps to think of workload intensity on a spectrum – not just how hard the actual work is, but how it hits you.
Budget building sits somewhere around a 6 or 7 out of 10 on a typical week, with predictable spikes around amendment cycles, and study startup phases where everything needs to be negotiated and signed ASAP. The pressure is real but it follows a pattern you can anticipate and you can recharge during the “off-season”.
Study Start-Up roles push that to an 8 or 9 during a site activation wave. The deadlines are hard, the dependencies are many, and the consequences of slipping are visible to everyone on the project. It is high-adrenaline work that some people thrive on and others find exhausting within a year.
On the other end, roles like Clinical Operations Analyst or Clinical Trial Financial Analyst tend to follow a steadier cadence – monthly and quarterly reporting cycles rather than launch countdowns. The work is still demanding but the pressure is distributed more evenly across the year.
Why does this matter for your decision? Because workload intensity affects everything downstream – burnout risk, work-life balance, and honestly how long you will stay in a role before looking for the next one. A higher salary in a role that runs at a 9 for six months a year is a different proposition than a slightly lower salary in a role that stays at a 6.
Salary Potential
Comparing salaries in clinical research is messy – it depends heavily on country, company type (CRO vs sponsor), and seniority. What matters more for a budget analyst evaluating a transition is understanding which direction leads to a higher ceiling – and which trades salary upside for stability.
Staying close to your financial roots – moving into Clinical Trial Financial Analyst or Outsourcing Manager roles – tends to offer strong stability and solid compensation that travels well across companies. You are unlikely to hit the highest ceilings in the industry, but you are also unlikely to find yourself unemployable in a downturn.
Moving into operational leadership – Clinical Project Manager, Study Start-Up Manager, eventually Director of Clinical Operations – is where you will find the highest ceilings. The tradeoff is real though: more responsibility, harder hours during peak periods, and a longer road to get there from a budgeting background.
Technical specialist roles like Regulatory Affairs or Biostatistics can command strong salaries, but as noted earlier, the barrier to entry is high and the path is narrower.
For actual numbers, Glassdoor and LinkedIn are useful for triangulating specific role titles at specific companies, but treat any single data point with skepticism – filter by country, company size, and years of experience before drawing conclusions.
As a side note, starting June 7th 2026, member states of the EU must transpose the Pay Transparency Directive into national law and companies must disclose salary ranges, which will make comparisons somewhat easier.
In the next article of these series I will go over specific roles and how they compare to a typical budget analyst.
Further Reading:
- ACRP Clinical Research Career Lattice
- Diverse Careers in Clinical Research (ICON plc)
- Key Clinical Research Roles (PharmiWeb)
- How to grow your career in clinical research
Resources like the ACRP Career Lattice and certifications through ACRP and SOCRA can help you identify competency gaps and demonstrate commitment to prospective employers during a transition. Both organizations also offer networking opportunities that are often more valuable than any certification alone.
Find out who I am and what I am fighting for.
If you agree, disagree, or think this is nonsense, email contact@clinicalbudget.com
If you’d like me to cover a specific topic, you can suggest it here.




Why Clinical Trial QC Feedback Often Sounds Harsher Than Intended