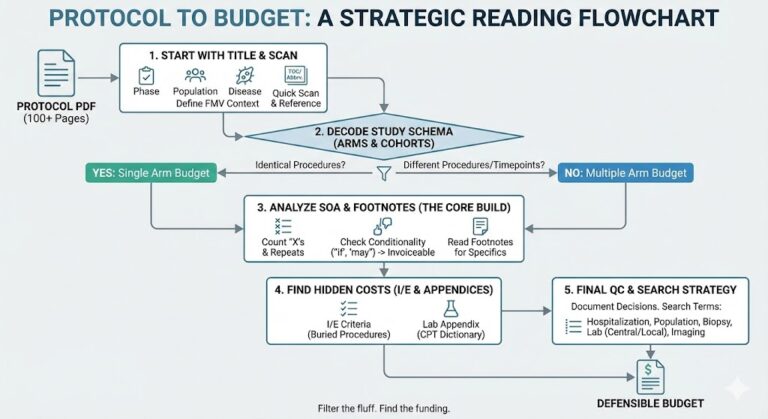
This guide is designed as a working reference for clinical trial bidding and pre-award budget development.
It is not meant to be read linearly. Use the search function to jump to the section you need.
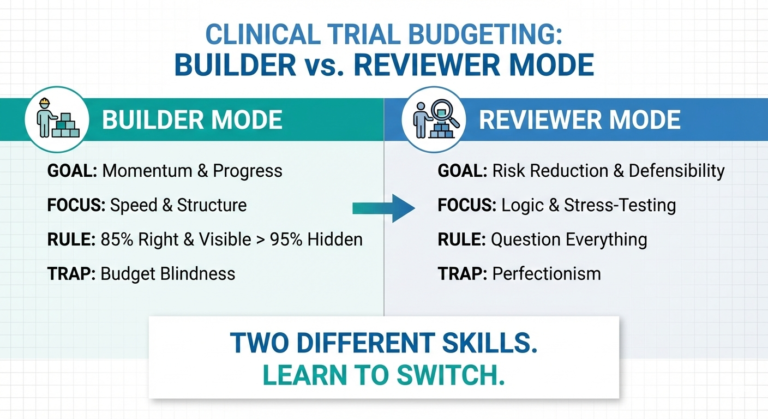
Bid building principles and how to make the QC-er’s job easier
The idea when creating a pre-award build, also known as a clinical trial budget bidding, is to provide an estimate of how much the study will cost, typically based on a preliminary version of the protocol. Both CROs and Sponsors utilize this in order to better plan their resources and priorities. As it is an estimate, not a final product, it is less detailed than a traditional budget build meaning that a visit grid might not be necessary, while also heavily relying on assumptions. It is essentially a trade-off between speed and accuracy.
The Simplified Representation
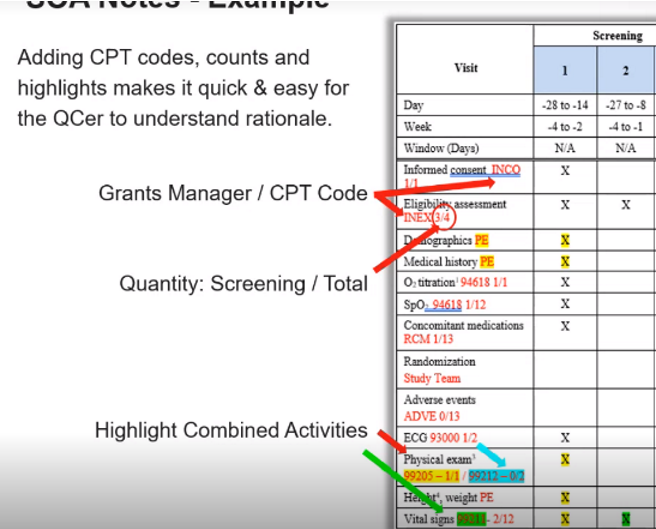
In a bid, we often don’t use a full visit grid. Instead, we count the total occurrences (frequency) of a procedure.
- The “Vertical” Count: When combining procedures, count vertically (per visit), for each timepoint individually. If three procedures are bundled into one visit fee, that counts as 1 unit for that visit.

The Red cells add up to 1; the green cells – to 2 (not to 6); the orange – to 2.5
- Assumed Ratios: If a procedure only happens for some patients, use a ratio (e.g., 0.5).
- Color Coding: Highlight bundled procedures in the same color so a reviewer can easily see your logic.
CPT Code Best Practices
- Always note the CPT code and the total frequency you’ve included.
- The “Overshoot” Rule: It is always better to slightly overestimate costs than to provide a lower number and have to explain a budget deficit later.
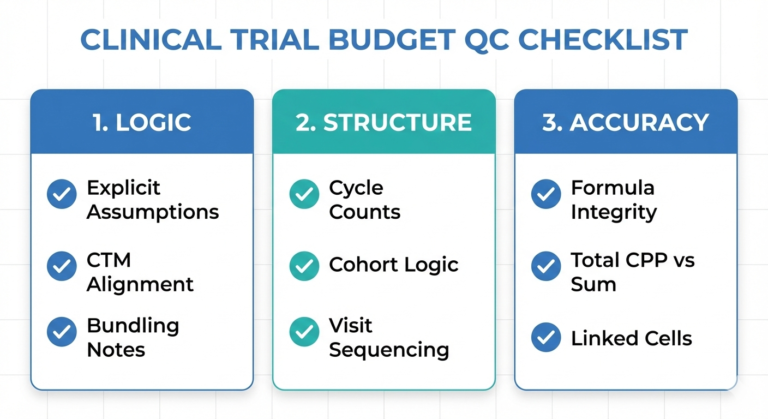
Below is an example of what a complete bid working file might look like:
Please note that the provided CPT codes throughout this guide refer to procedures in Medidata’s Grants Manager. Also be sure to take into consideration your employer/Sponsor’s specifics, as they can make a substantial difference.
Rules you apply to every bid (Cycles, Visits, Consent, Randomization)
Cycles: when the trial has cycles, carefully count the procedures multiplied by the number of repetitions. If the number of cycles is indefinite (i.e. Until something happens to the patient), assume a reasonable number and highlight this in the list of assumptions you are preparing. For example, we have “Day 1, Cycles 4+”, we need to add these procedures for Cycle 4 Day 1, Cycle 5 D1, C6D1 and so on, up to what the Grants Request Form specifies or a reasonable number (say 10).
Conditional visits:
- Unscheduled visit – if specifically added as a visit column in the Schedule of Activities (SOA), count it as 1;
- Early Termination (ET)/Early Withdrawal visit – even if it has been added separately as a specific visit, we should not count it as a separate visit because the drop-out rate will assume for it;
- End of Study/End of Treatment visit – to be added as a regular visit because it will be performed on each patient, no matter when they complete the treatment;
- ET/EOT – if combined, will be counted as 1 for the EOT part (see the statement above).
Optional Visit – if a visit is marked as optional, count each procedure timepoint as 50%.
Phone call visit – if a visit is indicated as a phone visit, add its respective procedures and then add a SC fee on top of that, PIs are usually not involved, so their fee might not be needed.
At Investigator’s discretion/as clinically indicated – add 0.25 to the total frequency of the item to cover occasional usage. For example, if there is a mandatory ECG at Screening and then “if clinically indicated” for the next 10 visits, add it with a total quantity of 1.25 – 1 from the Screening and 0.25 for the total of follow-up.
Always add Informed Consent, even if it is for some reason missing from the Schedule of Activities. If we have a pediatric study, we need to add both INCO and PNCO as 1, since a child consent form is needed, as well as their parents. When we have a mixed study of adults and kids (INCO (adult patient consent form) + PNCO (child consent form)) – we count INCO as 1 and PNCO as 0.5. The assumption here is that only half of the population will need the PNCO code, while everyone needs the INCO code. Be sure to check if your FMV tool has separate codes for pediatric exams i.e. physical exams as XXXXX-PD.
Randomization – we don’t need to pay for it usually, assumed a part of the Study Coordinator (SC) Fee
Labs – think about purpose and what can be grouped together
Local Lab vs Central Lab
With Local labs samples are taken and analyzed at the site, so everything should be paid in full. When the study uses a Central Lab, a lab off site performs the tests and only the retrieval of the samples and the transport to it are paid to the institution and included in the budget. The Sponsor has a separate agreement with the Central Lab where the details of the cost of the tests is specified.
The email request can contain the info. If it is Local – we count as per standard
Central Labs, use a specific code (80299 – Sample) that only reflects the sample collection and another code is used for shipping (99000) to the lab.
It is possible for tests to require a single sample regardless of the type of labs. For example Hepatitis B and C, might have separate lines in the SOA but could be tested via a single serum sample (Serology). In those cases, we need to count it once per visit. Note that, if Hematology and Urinalysis are combined under Safety Assessments and we have a Central Lab involved, we will need 2 codes – one for the blood sample, one for the urine sample. Think about what can be united and justify your decisions in the assumptions file.
Types of Samples – check your FMV tool for specific codes Skin, Tissue, Bile, Bone, Breath, CSF (Cerebrospinal fluid), Feces and Nails.
DNA Sample – taken with a sample (80299) code. The assumption is that this will be kept for years to come and it should be added with a separate line. Note that this activity might not apply for all patients.
PK Sample – medicine concentration in the blood and how much time it is needed to process the drug. Always a separate line from other blood samples . Almost always Central Lab, if it is Local, there is a special code – 80299-AN
Blood Tests:
Coagulation – when coagulation is Local lab, but it’s not specified whether it should be aPTT (85730), INR or PT (or all three), use the code for PT (85610) + the code for INR (NC108);
Serology (HIV (86703), HCV (86803), and HBsAg (87340)) – Each of this tests needs to be added separately;
Lipid panel (80061) – Separate code from the Local Blood Sample;
C-reactive protein (86140) – Separate code from the Local Blood Sample;
Biochemistry Local – NC125 for 13 and more markers // NC124 for 12 and less. We can typically use NC125, since it is more expensive. Central – we use 80299. If fewer than 5 chemistries are being assayed, break out each one individually.
Most common ones include 80061 – Lipid panel, includes Cholesterol (82465), Triglycerides (84478), High Density Lipoprotein (HDL, 83718) (and Low Density Lipoprotein (LDL, 83721)).
Safety Lab – When a protocol does not clearly outline which safety labs will be performed, assume that safety labs include Hematology with differential (85025), Serum Chemistries, 13 or more (NC125), and urinalysis with microscopy (81002).
Pregnancy – we typically assume at 50%, pay attention if it is Urine or Serum. Typically unless otherwise specified the first one is Serum, the rest are Urine. Always a Local Lab and always Invoiceable. Serum – 84702; Urine – 81025
Antibodies to Study Drug or a comparator drug/Immunogenicity – Consider using code NC045 – Antibody to Specific/Study Drug or Comparator’.
Clinical assessments – often bundled, unless repeated
Physical Exam – the initial visit is always more expensive. It could also be called Comprehensive or complete (99213), Focused (99231) or Abbreviated (99212). Typically a Physical Exam includes Vital Signs measurement and Height/Weight, which could be different rows in the SOA. Vital Signs, when not a part of a physical exam or if we have more occurrences at a single visit than physical exams, have a different code – 99211.
In case the study is fully pediatric, the following codes should be assumed: 99205-PD, 99211-PD, 99212-PD.
Clinical data collection – We can combine this with a physical exam of the Study Coordinator fee.
Prior and Concomitant Review – code RCM, may occur as Concomitant Medication (other medication that the patient takes).
Adverse Events – code ADVE, could be marked as Serious Adverse Events (SAE).
Demographics – rarely a separate line, typically combined with Medical History.
Procedures – Imaging and Biopsies as main cost drivers
ECG – be careful if performed more than once on certain visits; 93000, may occur as 12-lead ECG.
6MWT – 6-minute walk test (94618)
MRI/CT = Imaging – could be different areas of the body and each type of scan has a different code + there are additional codes for scans with or without contrast material. Unless otherwise specified you should use the with contrast option, as it costs more. MRI is always more expensive than CT. Unlisted (76498) if the area is not specified. If we have a MRI/CT row – we divide it by 2 and add a code once for both MRI and CT.
CT CAP – CT Chest, abdomen and pelvis (NC173)
RECIST stands for Response Evaluation Criteria in Solid Tumors – Add it separately (WTR), but only when the scans are for solid tumors.
Tumor Specimen – we have to find out if it is either Fresh or Archival Biopsy – When it’s either fresh or archival biopsy, include the fresh biopsy at 50% and add the complex biopsy handling code at 100% (NC171). This way, both the handling and shipping of the fresh biopsy and the handling and shipping of the archival sample are accounted for.
Note: If it’s specified in the protocol that the biopsy will be analyzed locally, then do not add complex handling, and add the relevant surgical pathology code instead. If not specified, we can assume that the samples will be analyzed centrally. Also note that unlisted biopsies are relatively cheap, so we might have to list the code for a specific biopsy, to cover the expenses.
Since Biopsies always require an additional informed consent, we need to add it to the count of INCO and explain why we are adding it. When we don’t have any specifics – 88305 – Level IV Surgical Pathology (used for most biopsies, and bone marrow aspirate).
Endoscopy/colonoscopy – there are more than 100 to choose from, so you can discuss with your team which are the most common ones. The table below can serve as a guidance.
Ulcerative Colitis and Crohn’s Disease
| Endoscopy/Colonoscopy Codes | Biopsy Code | Biopsy Handling |
| 45380(Colonoscopy with biopsy, single or multiple) | N/A (Included in 45380) | SimpleBiopsy Sample Handling (inc staining/prep of slides reviewed elsewhere) Simple: 1 to 2 task(s)ComplexBiopsy Sample Handling (inc staining/prep of slides reviewed elsewhere) Complex: 3+ task(s) |
Eosinophilic Esophagitis
| Endoscopy/Colonoscopy Codes | Biopsy Code | Biopsy Handling |
| 43239(Esophagogastroduodenoscopy (EGD), Flexible, Transoral; with biopsy, single or multiple) | N/A (Included in 43239) | SimpleBiopsy Sample Handling (inc staining/prep of slides reviewed elsewhere) Simple: 1 to 2 task(s)ComplexBiopsy Sample Handling (inc staining/prep of slides reviewed elsewhere) Complex: 3+ task(s) |
IP & study management – oversight, handling, and admin effort
Investigational Product (IP) Administration:
- Oral/Cream – the patient can administer it himself.
- Injections – need to be added in your budget, since a specialist needs to administer them – different code depending on the type of injection. The most common type is intramuscular, subcutaneous (96372).
- Infusions are expensive and the price is determined by the length. If we have 3 hours – code for 1 hour (96365) + codes for each additional hour (96366). Intravitreal Injection – 67028.
In-clinic IP Administration – if we have an oral drug, we can assume that it is a part of the SC Fee, so we do not need to add an additional code. We can mark the code as “Part of SC Fee” in our assumptions.
ISRS – we can use this code if the site of the drug administration/application (this includes Infusion) needs to be monitored for a reaction
IР accountability and return and IР compliance review can be added as CUSD. Collection of Unused Study Drugs is also marked as CUSD and is typically taken only once.
TOHM – Training On Home Monitoring Devices (eg Blood Pressure Monitoring Devices, Glucose Monitoring Devices, E-Diary). We use this code when we have a device that the patient must be trained on. Note that it might not be listed in the description but the code might still be appropriate.
Diaries – Diary Card Provided – DPSD; Diary Card Collected – CPSD; Diary Card Reviewed – RPSD.
Questionnaires – there are a lot of specific codes for all the types of questionnaires. One example is DIMQ Disease Impact Questionnaire – this is the generic code. When trying to identify a comparable Questionnaire or Assessment two of the most important factors to keep in mind that will affect cost and complexity, is the administration time and whether it is Self-Reported, an Interview, a Clinical Assessment or a Standardized test.
Training Evaluation Questionnaire (TEQ) can be used for activities such as Pregnancy avoidance guidance.
Special study types
Home Healthcare (HHC) – Two options:
- If it’s explicitly stated in the Protocol/Grant Request Form that the HHC visits will be done by a vendor, do not include the procedures for these visits in the bid. Do include PI and SC fees for them, though, as the sites still have oversight and need to review/record the information provided by the HHC vendor.
- If it’s not clear whether the HHC visits will be done by a vendor or by the site, include the procedures for these visits at 50% and the PI and SC fees at 100%.
Observational Studies
Observational studies (sometimes marked as “non-interventional study”) are one of the easiest to do and there are only a limited number of activities that you have to add.
Very carefully go through the Protocol in order to understand, if the study is going to be entirely observational or, if some procedures are still to be included and paid by the Sponsor.
- If the study is entirely observational, the following is normally included:
- Activities – Informed consent, Inclusion/exclusion criteria, EDC (electronic data capture)
- Other Direct Costs – PI fee, SC fee, Start-up fee, Archiving fee
If the study is not entirely observational, additional items, for example questionnaires, may need to be added on top of the above.
Many protocol descriptions are intentionally vague at the bid stage. The goal is not perfection, but defensible assumptions.
Default rules when bid building
- When unclear, if it will occur or not → include at 50%
- When lab unclear → assume Local
- When type of procedure is vague → choose more expensive reasonable code
- When in doubt → document assumption
Feel free to use this clinical trial bidding cheat sheet in your next assignment:

Find out who I am and what I am fighting for.
If you agree, disagree, or think this is nonsense, email contact@clinicalbudget.com
If you’d like me to cover a specific topic, you can suggest it here.