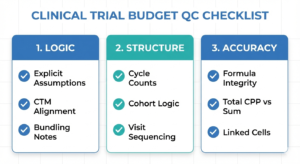
Why Clinical Research Budget Builders Feel Behind (Even When They’re Doing Fine)

Many clinical research budget builders feel behind or inadequate, even when they’re technically competent. This isn’t a flaw in you - it is the result of delayed feedback loops, ambiguous protocols, and training that teaches how to build, not how to think. In this article, we explore why these roles feel harder than they should and how to shift your mindset and workflow to build confidence, reduce anxiety, and improve performance.