Protocol Amendments in Clinical Trials: Impact, Process, and Budget Considerations

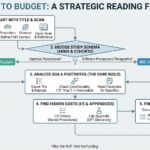
Protocol amendments are an inevitable reality of clinical research, not a reflection of budgeting errors. This guide explores how to distinguish between simple and complex amendments, manage the impact on site contracts, and execute a methodical "Delta Analysis" to keep your study on track.