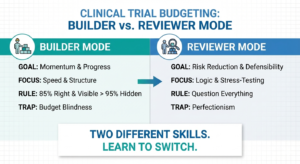
Clinical Trial Budgeting: Mastering the Builder vs. Reviewer Mindset
Building and reviewing a clinical trial budget require two completely different modes of thinking. Builders translate ambiguity into momentum, while reviewers stress-test assumptions and look for hidden risk. When we try to apply both lenses at once, cognitive overload sets in and mistakes feel personal. Mastering the switch between builder and reviewer mindset is not about talent - it is about learning how to think in stages.